Introduction
Material and Methods
1. Sample preparation and descriptions
2. Experimental design
3. Oxygen uptake rate evaluation
4. Analytical methods
5. Statistical analysis
Results and discussion
1.General characteristics of screened and decanter-separated pig slurry solids for composting
2. Oxygen uptake rate
Conclusion
Introduction
The management of livestock manure has become a significant environmental and public health concern due to the rapid intensification of global animal production systems. Pig manure is one of the most challenging types of animal waste to manage due to its high organic matter and nutrient contents, as well as its potential to generate odors and emit greenhouse gases if improperly managed (Bernal et al., 2009). These characteristics underscore the need for effective treatment strategies that can mitigate environmental impacts while stabilizing organic matter and nutrients in manure. Accordingly, sustainable and biologically viable treatment methods, such as aerobic composting, have gained increasing attention as practical approaches for converting raw manure into stabilized organic fertilizer (Zhang et al., 2019).
Composting is a natural process that happens with the help of microbes. It breaks down organic matter in the presence of oxygen, which leads to the creation of stable humic substances that can make soil more fertile. Microorganisms oxidize organic materials during composting, releasing carbon dioxide, water, and heat while also turning organic waste into a stable, mature product (Haug, 2018; Richard, 2002). Aerobic composting offers several advantages over anaerobic digestion methods, including faster organic matter degradation, reduced odor emissions, and enhanced hygienization through the activity of thermophilic microorganisms (Liang, Das, & McClendon, 2003).
A lot of physicochemical and biological parameters affect the efficiency and quality of the composting process. Particularly, temperature, moisture content, porosity, oxygen availability, C/N ratio, and particle size are recognized as key variables that directly influence the microbial activity and compost stability (Richard, 2002; Ahn et al., 2007). The final quality of composting product needs the proper adjustment of these parameters might be accelerate composting process. For instance, Bernal et al. (2009) stated that keeping a balanced C/N ratio (typically between 25:1 and 35:1) is crucial for optimal microbial metabolism and minimizing nitrogen losses. Similarly, adequate aeration ensures sufficient oxygen supply, which is critical for sustaining aerobic conditions and preventing the onset of anaerobic zones that lead to odor formation and process inhibition.
In pig slurry treatment systems, mechanical pretreatment processes such as screening and decanting are commonly applied to separate raw slurry into solid and liquid fractions. These pretreatment processes reduce volume, enhance handling, and allow for targeted treatment or reuse of separated fractions. Nevertheless, there is limited understanding of the biodegradability of these separated manure components, especially in the context of their potential use in composting. Since the efficiency of composting is contingent on the microbial degradability of the feedstock, evaluating the biological stability of screened and secondary decanter-separated solids is essential for designing efficient composting strategies (Muller et al., 2002).
Oxygen uptake rate (OUR) measurements are widely used to evaluate the biodegradability of composting feedstocks and to assess the suitability of composting conditions based on microbial respiration activity. OUR is a respirometric indicator that quantifies the rate of oxygen consumption associated with microbial degradation of organic matter (Tiquia et al., 1996). Accordingly, OUR provides insight into the biological stability and maturity of composting materials, as well as their potential performance during the composting process (Barrena et al., 2009). Higher OUR values generally indicate elevated microbial activity and lower biological stability, reflecting materials that are either in the early stages of composting or characterized by high biodegradability.
Due to the limited research assessing the composting potential of separated manure fractions based on respiration rate, this study aims to evaluate the biodegradability of primary screened and secondary decanter-separated solids obtained from an animal manure management facility using the oxygen uptake rate (OUR) method. While previous studies have primarily focused on the physicochemical characteristics of manure rather than microbial respiration, the main objective of this study is to assess the suitability of these solids as composting feedstocks by employing the OUR method. Additionally, the study investigates the effects of different mixing ratios between these fractions and varying levels of sawdust addition as a bulking agent on composting conditions. By comparing microbial respiration under these combinations, this research seeks to provide a scientific basis for optimizing composting strategies and enhancing manure valorization in swine production systems.
Material and Methods
1. Sample preparation and descriptions
The pig manure samples used in this study were collected from an animal manure management facility associated with a commercial swine farm with approximately 7,000 pigs, located in Pyeongchagng-gun, Republic of Korea. The solids separation process at the facility consists of a primary screen and a secondary decanter, and the solids obtained from each process were used as experimental materials. In addition, a mixture of both the primary screened and secondary decanter solids was collected to determine the most suitable substrate for the composting process. The collected samples were placed in sealed containers, transported to the laboratory, and stored at 4°C until analysis. The general characteristics of the substrate are presented in the Table 1 and Figure 2 and 3 below.
2. Experimental design
The experimental design comprised seven test groups: T1 (primary solid parts separated by screen without mixing), T2 (secondary solid parts separated by decanter without mixing), T3 (mixture of both primary screened + secondary decanter parts+woodchips), T4 (primary + secondary mixture (1:1, based on the wet weight) + sawdust), T5 (primary + secondary mixture (1:1, wet weight)), T6 (primary + secondary mixture (1:2, wet weight) + sawdust), and T7 (primary + secondary mixture (2:1, wet weight) + sawdust). T1 and T2 served as sole treatment groups representing primary screened solids and secondary decanter-separated solids, respectively. T3 was prepared as a composting raw material by mixing woodchips with separated solids intended for input into the facility's composting process; the sample provided by the facility was used as-is without additional mixing. To evaluate the effects of mixing ratios and bulking agent addition, treatments T4-T7 were prepared in the laboratory by weighing T1 and T2 based on wet weight basis and manually mixing them in a plastic bag for 10 minutes to ensure homogeneity.
3. Oxygen uptake rate evaluation
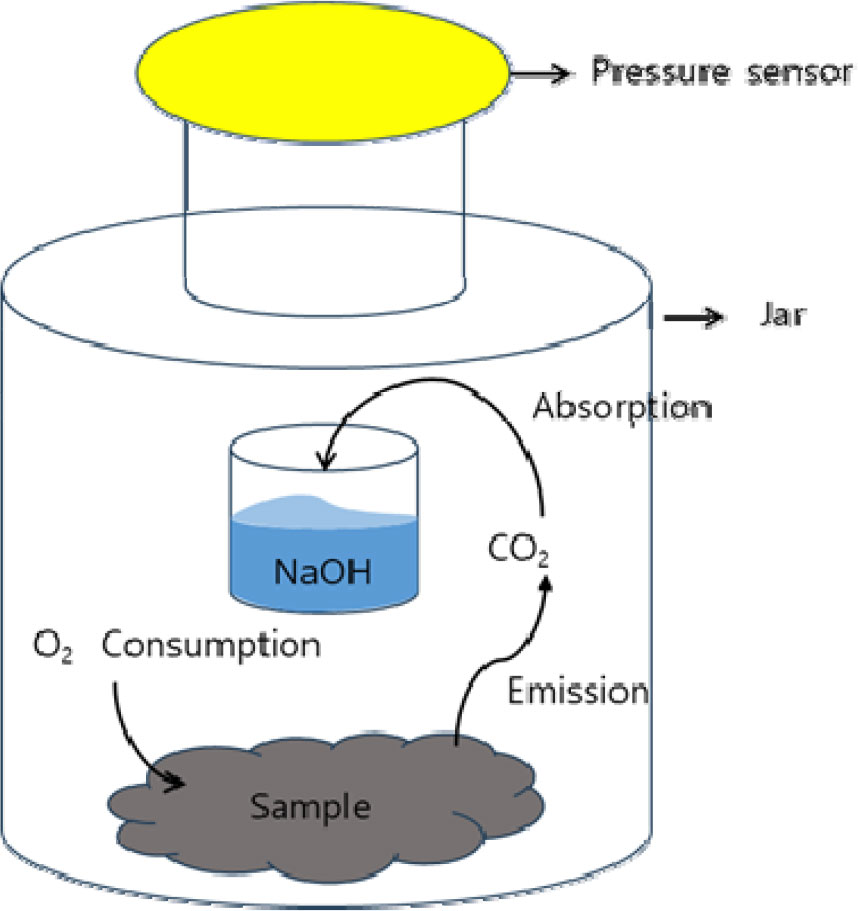
The oxygen uptake rate was measured by a pressure sensor method (OxiTop system, (Ahn, Richard, & Glanville, 2008; Sadaka, Richard, Loecke, & Liebman, 2004) ) at different moisture levels. The moisture contents of each treatment group were ranges between the 53% to 68% as presented in the Table 1. The collected sample was properly broken down the wood parts to the appropriate particle size and subjected to incubate in chamber at 37°C prior to oxygen uptake measurement conducting. Then about 10-15 g of the well-mixed sample add to the container 1 L volume jar a pressure sensor heads with data-logging function (OxiTop-C WTW, Weiheim, Germany), and a plastic cup for NaOH solution as shown in Figure 1, and disperse the sample evenly to prevent clumping. Aerate gently for 5-10 min to ensure oxygen saturation (DO ≥ 5 mg L-1). The solution of NaOH was prepared by 1 M NaOH and 40 ml of NaOH solution was added into the plastic tube and placed in the 1 L volume jar. The jars were tightly sealed and incubated at 37°C for 7 days. All measurements were done in triplicate. The oxygen consumption was determined according to the following equation (Ahn et al., 2008). A controller (OxiTop OC 110 WTW, Weiheim, Germany) is used to collect data from the pressure sensor data loggers. Company supported Software (Achat OC, PC communication software version 2.03) is used to download the data from the controller to a spreadsheet.
Table 1.
Initial and final moisture content and volatile solids of the experimental groups (Mean ± Standard deviation).
Notes: N/A: (Not Available), T1: (primary solid parts separated by screen); T2: secondary solid parts separated by decanter); T3 (mixture of primary + secondary parts+woochips); T4: (primary + secondary mixture (1:1, wet weight) + sawdust); T5: (Primary + secondary mixture (1:1, wet weight); T6: (Primary + secondary mixture (1:2, wet weight) + sawdust); and T7:(primary + secondary mixture (2:1, wet weight) + sawdust)
where
O2 is the oxygen consumption (mg/g vs·d)
∆P is the gap between the maximum and final pressure (hPa),
V is the jar volume (m3),
T is the incubation temperature (°K),
t is the incubation time (h),
W is the weight of sample (g).
4. Analytical methods
The moisture content (MC), total solids (TS) and volatile solids (VS) were determined using the standard method suggested by the American Public Health Association (APHA, 2017), wherein samples were oven-dried to a consistent weight at 105°C and subsequently ignited for 8 hours at 550°C. The pH and electrical conductivity (EC) of the samples were measured utilizing a digital pH meter with a combined glass electrode (Thermo Scientific, Orion 4 Star pH/Conductivity Benchtop Meter). The elemental composition of treatments where Carbon, and nitrogen (C/N) elemental compositions were assessed by pulverizing dry samples and assaying 0.50 g powdered samples in a Vario elementary cube system (Elemental Analyzer System).
Free air space (FAS) of organic materials was determined by the following equation reported by the (Ahn, Sauer, Richard, & Glanville, 2009).
Where MC is the moisture content (decimal, weight basis), VS is volatile organic solid (decimal, dry basis), DM is dry matter (decimal, weight basis), Pwb is wet bulk density of sample (kg/m3), Pw is density of water (1,000 kg/m3).
4. Statistical analysis
Statistical analyses were performed using Stata version 16.1 (Stata Corp LLC, College Station, TX, USA) to evaluate the significance of differences among treatment groups. One-way analysis of variance (ANOVA) was employed to assess variations in all measured parameters. When significant differences were detected (p < 0.05), post-hoc multiple comparison tests were conducted using Tukey’s HSD to determine specific group differences. All data were expressed as mean ± standard deviation (SD).
Results and discussion
1.General characteristics of screened and decanter-separated pig slurry solids for composting
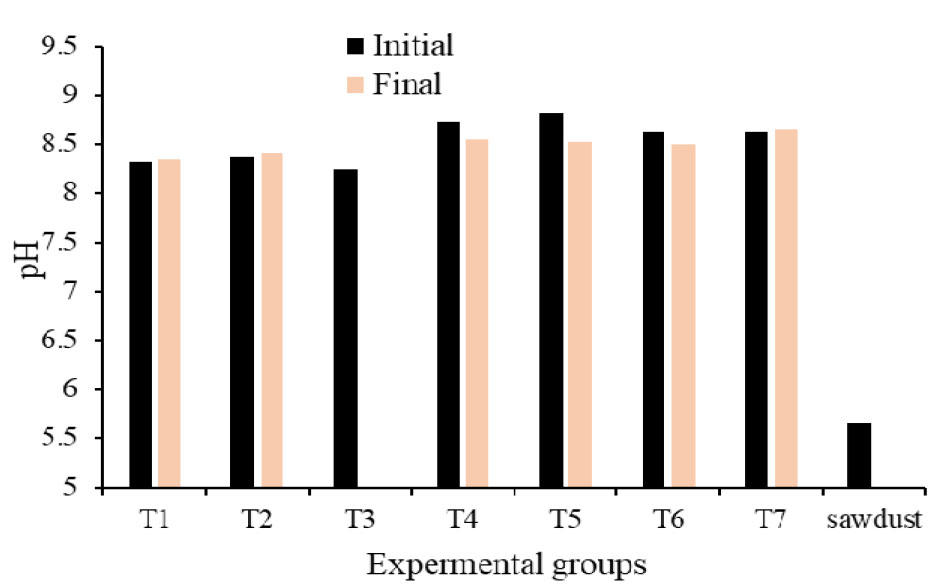
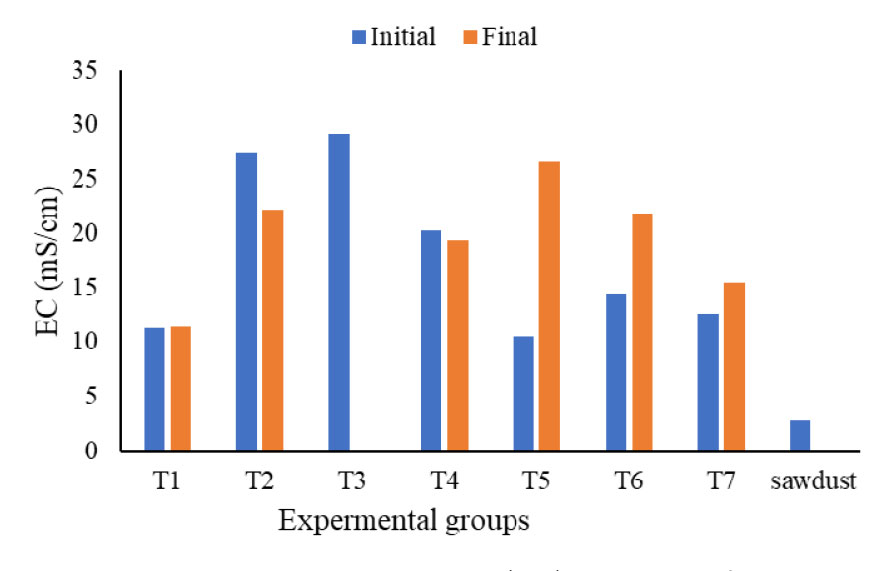
The initial and final moisture contents and volatile solids are summarized in Table 1. The moisture contents of initial substrate ranged from 55% to 70%. The highest moisture level was observed in T2 (secondary solids separated by decanter), which correlated with a higher oxygen uptake rate, suggesting that increased moisture within this range may have facilitated greater microbial activity. This study aligns with the finding of Ahn et al. (2008), who reported that the oxygen uptake rate increases proportionally with the moisture content of the bulking agents. Although a universally optimal moisture level for various composting materials does not exist, higher moisture content within the range of the material’s water holding capacity tends to increase the oxygen uptake rate (OUR), reflecting enhanced microbial activity (Li et al., 2008; Liang et al., 2003). pH is another parameter that may play an important role in the oxygen uptake rate of substrates, and the results are presented in Figure 2. Apart from sawdust (pH 5.6), the initial and final pH values of all substrate treatment groups ranged from 8.2 to 8.8. The concentration of electrical conductivity (EC) of the initial and final experimental groups are presented in the Figure 3. Electrical conductivity serves as an indicator of salinity within the organic matter; depending on the concentration, these dissolved salts can either stimulate microbial metabolism or inhibit activity through osmotic stress.
The biodegradability of organic matter during oxygen uptake rate (OUR) measurement is highly dependent on the contents of carbon to nitrogen ratio (C/N). As presented in the Table 2, the C/N ratio of sawdust was higher, close to 299 compared to the T2 (secondary solid parts separated by the decanter) which was 9.6. The rate of microbial respiration is directly influenced by nutrient balance. In this study, substrates amended with sawdust showed a lower oxygen uptake rate compared to those without sawdust. This suggests that the addition of sawdust into the other substrates assumed to be increase the carbon-to-nitrogen (C/N) ratio beyond the optimal range, potentially creating a nitrogen limitation that might be slow microbial metabolic activity (Ahn et al., 2008). Substrate porosity significantly affects composting efficacy, as optimal physical conditions for air distribution must be preserved throughout the process as presented in the Table 2.
Table 2.
Bulk density, free air space (FAS) and carbon to nitrogen ratio (C/N) of comopost feedstocks used in this study.
Porosity greater than 50% causes the composting pile to remain at a low temperature because energy lost exceeds heat produced. Too little porosity leads to anaerobic conditions and odor generation. The percentage air-filled pore space of composting piles should be in the range of 35-50% (Barnel et al., 2009). In this context, the free air space of the present study varied, with T2 (secondary solid parts separated by a decanter) ranging between 30 and 50%, close to 45%, which indicated that the space was appropriate for the activity of microbial processes during composting process.
2. Oxygen uptake rate
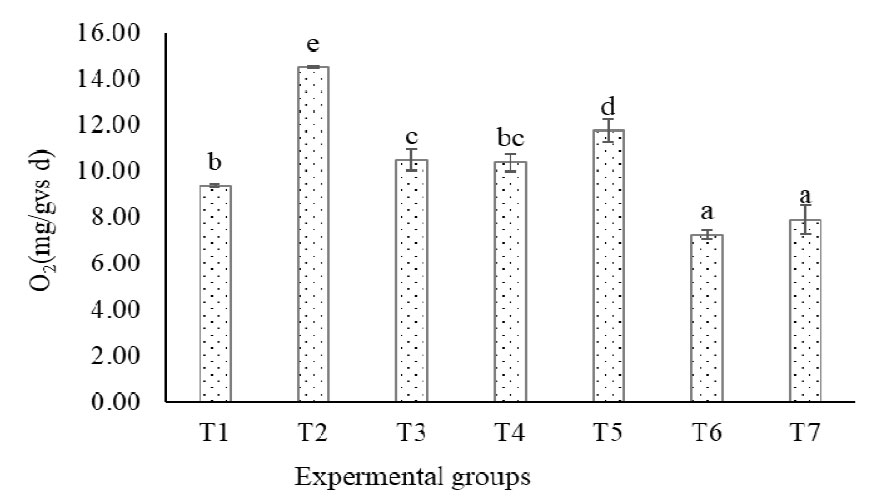
The amount of oxygen consumed by the aerobic microorganisms during the oxygen uptake rate measurement of screened and decanter-separated pig slurry solids is presented in the Figure 2 and Table 3. The oxygen uptake rate of the treatment groups ranged between the 7.1 to 14.2 mg O2/g VS·d. The highest oxygen uptake rates were observed in the T2 (secondary decanter-separated pig manure solids), while the lowest oxygen uptake rate recorded in the T6 (a mixture of primary and secondary separated manure solids combined with sawdust). Following T6, the T5 (a mixture of primary and secondary separated manure at a 1:1 wet weight ratio) showed relatively high OUR of 11.5 mg O2/g VS·d (p < 0.05). However, no significant difference was observed between T6 and T7, as well as between T1 and T3.
Table 3.
Comparison of daily oxygen uptake rates (mg O2/g VS・d) of screened and decanter-separated pig manure solids.
It is important to note that although the controlled OUR test conditions (37°C) differ from the dynamic temperature fluctuations of full-scale composting, this respirometric analysis provides a indicator for comparing the intrinsic early-stage biodegradability of feedstocks. Accordingly, the presence of higher oxygen uptake rate (OUR) in this study is indication of the presence of easily biodegradable organic matter, which stimulates microbial activity and leads to increased oxygen consumption. In contrast, lower OUR values, such as those observed in substrates combined with sawdust, suggest the presence of recalcitrant materials that reduce microbial activity and oxygen demand. Although sawdust is commonly used as a bulking agent to improve composting performance by adjusting moisture content and enhancing free air space when these parameters are suboptimal, its addition may adversely affect microbial respiration when moisture and aeration conditions are already within an appropriate range. Under such conditions, the incorporation of sawdust can dilute readily biodegradable substrates and introduce lignin-rich recalcitrant material, thereby reducing oxygen uptake rates. The finding is aligned with previous studies, where oxygen uptake measurements of substrate combined with sawdust and rice hulls demonstrated noticeably different rates. Beef cattle combined with sawdust displayed a low OUR of 13.2 mg O2/g VS·d, while beef cattle manure combined with rice hulls showed a higher oxygen uptake rate (OUR) of 33.3 mg O2/g VS·d (Kim, Lee, Won, & Ahn, 2016). Similarly, (Ahn et al., 2008) reported that sawdust showed one of the lowest oxygen uptake rates among common bulking materials, attributable to its high C/N ratio and substantial lignin content. These findings, together with the present results, emphasize that the effectiveness of bulking agent addition depends strongly on the initial physicochemical properties of the composting substrate. Another possible reason is the favorable environment created for microorganisms due to the free air space within the substrate. Optimal free air space typically ranges between 30% and 50% (Külcü & Yaldiz, 2014), and T2 (secondary solid parts separated by decanter) fell within this range at approximately 45%, while free air space of sawdust alone was 82% (Table 2).
Conclusion
This study evaluated the biodegradability of screened and secondary decanter-separated pig manure solids using the oxygen uptake rate (OUR) method, focusing on their early-stage biodegradability and oxygen demand characteristics under standardized conditions relevant to feedstock preparation prior to composting. The results demonstrated that secondary decanter-separated solids (T2) and the 1:1 mixture without sawdust (T5) exhibited higher OUR values compared to other treatments, indicating greater early-stage biodegradability and oxygen demand. In contrast, mixtures containing sawdust showed significantly lower OUR values, likely due to a combination of factors such as an elevated carbon-to-nitrogen (C/N) ratio, a higher proportion of lignocellulosic (recalcitrant) carbon, and dilution of readily biodegradable substrates. These compositional changes can collectively reduce apparent microbial respiration in short-term OUR assays, depending on moisture content and structural conditions.
The findings also underscore the role of physicochemical properties such as moisture content, bulk density, and free air space (FAS)—in governing microbial oxygen consumption. Variations in these parameters among treatments likely contributed to the observed OUR differences. The relatively favorable moisture and FAS conditions observed in T2 may have supported enhanced aerobic degradation during the OUR test, whereas sawdust-amended substrates introduced structural and compositional changes that may limit microbial activity unless moisture and aeration are carefully managed.
Overall, the study demonstrates the utility of OUR as a diagnostic screening tool for assessing early-stage biodegradability and oxygen demand, and for informing feedstock selection and early-stage process control, rather than for directly evaluating final compost maturity or stability. Accordingly, the results offer preliminary decision-support information for optimizing substrate preparation and aeration strategies prior to composting. These insights can enhance the efficiency of manure management systems by guiding the formulation of feedstocks with higher early-stage biological activity in swine production systems. Future research should validate these findings under pile- or reactor-scale composting conditions by linking OUR values to temperature evolution, oxygen availability, odor emissions, and compost maturity and stability indices to confirm full-scale applicability.